| Catalogue number |
C104687 |
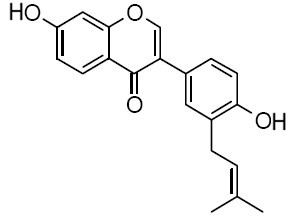
| Chemical name | Neobavaisoflavone |
| CAS Number | 41060-15-5 |
| Synonyms | 4’,7-Dihydroxy-3’-(3-methyl-2-butenyl)isoflavone; 7-Hydroxy-3-[4-hydroxy-3-(3-methyl-2-buten-1-yl)phenyl]-4H-1-benzopyran-4-one |
| Molecular Weight | C20H18O4 |
| Formula | 322.4 |
| Purity | 98% |
| Physical Description | Cryst. |
| Solvent | Chloroform, Dichloromethane,DMSO |
| Storage | Stored at 2-8°C, Protected from air and light, refrigerate or freeze |
| Applications | Neobavaisoflavone was isolated as a DNA polymerase inhibitor.
In the present study we investigated in vitrothe anti-inflammatory activity of neobavaisoflavone. Macrophages play an important role in inflammation through the release of inflammatory mediators involved in the immune response. Inappropriate and prolonged macrophage activation is largely responsible for the pathology of acute and chronic inflammatory conditions. Neobavaisoflavone significantly inhibited the production of reactive oxygen species (ROS), reactive nitrogen species (RNS) and cytokines: IL-1β, IL-6, IL-12p40, IL-12p70, TNF-α in LPS+IFN-γ– or PMA– stimulated RAW264.7 macrophages.
Neobavaisoflavone concentration-dependently promoted osteogenesis in MC3T3-E1cells, demonstrated by notable enhancement of alkaline phosphatase (ALP) activity, increase of bone-specific matrix proteins expression including type I collagen (Col-I), osteocalcin (OCN) and bone sialoprotein (BSP), and formation of bone nodules. Neobavaisoflavone up-regulated the expression of runt-related transcription factor 2 (Runx2) and Osterix (Osx), the bone-specific transcription factors participating in regulation of bone marker genes expression. the osteogenic activity of neobavaisoflavone might probably act through activation of p38-dependent signaling pathway to up-regulate the mRNA levels of Runx2 and Osx then stimulate bone matrix proteins expression. Neobavaisoflavone might be a potential anabolic agent to treat bone loss-associated diseases.
|
| References | 1. Bull. Chem. Soc. Japan, 1978, 51(8), 2398-2400.
2. Journal of Chromatography B, 2005, 821(1), 67-74.
3. J. Nat. Prod. , 1998, 61(3), 362-366.
4. Molecules, 2011, 16(5), 3701-3712.
5. Phytomedicine, 2012, 19(6), 551-561.
|
| Guestbook |
|
The packaging of the product may have turned upside down during transportation, resulting in the product adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling.
| Size | Price(USD) | Discount |
| 5mg | Inquiry | N/A |
| 10mg | Inquiry | N/A |
| 25mg | Inquiry | N/A |
Orders can be placed by Emails. All orders received will be shipped in the next day if the stock is available.
To place an order, please provide the following information.
1) Your name and telephone number
2) Purchase order number
3) Product number, package size, description, and quantity
4) Shipping and billing addresses
Sent to your order to our email: info@coompo.com
If you have any questions about discounts or dealer discount, please send us a message. We will be glad to help.